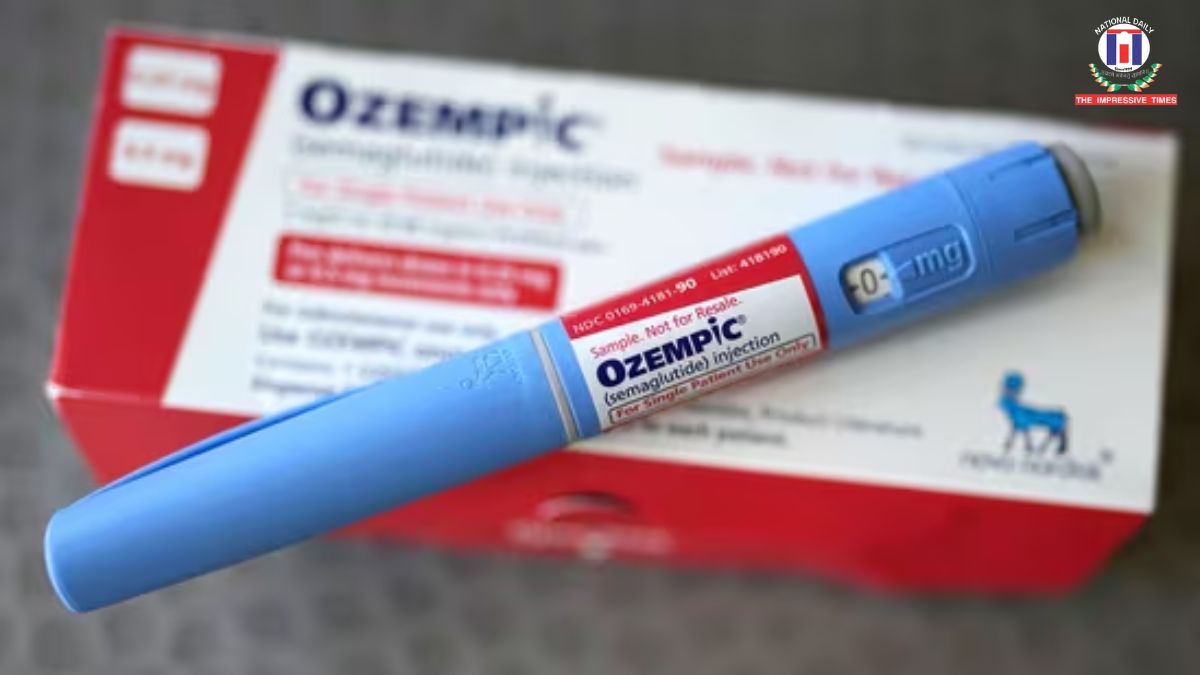
Ottawa- In a significant development for diabetes care, Health Canada has approved the first generic version of Ozempic, originally developed by Novo Nordisk. The newly approved version is manufactured by Dr. Reddy’s Laboratories, marking a major step toward more affordable treatment options.
The approval is expected to intensify competition in the market for GLP-1 therapies, which are widely used for managing type 2 diabetes and have also gained popularity for weight loss. Since its launch in 2018, Ozempic has become a blockbuster drug globally, but its high cost has remained a concern for many patients.
With generic versions entering the market, prices are likely to fall significantly. In Canada, generic medicines are typically 45% to 90% cheaper than branded drugs, making treatment more accessible to a broader population. Analysts say the Canadian rollout could serve as a test case for how generics compete with branded peptide-based therapies.
The move follows similar developments in India, where copycat versions of semaglutide were introduced after certain patent protections expired. This has already prompted price cuts by Novo Nordisk for Ozempic and its obesity drug Wegovy in that market.
Meanwhile, competition in the GLP-1 segment is heating up, with rival companies like Eli Lilly gaining ground through drugs such as Mounjaro and Zepbound.
Health Canada is also reviewing multiple additional applications for generic semaglutide, suggesting that more low-cost alternatives could enter the market in the coming months.















No Comments: